PRISMA 2020 (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) is the current reporting standard for systematic reviews. The 27-item checklist and the four-phase flow diagram together specify what a systematic-review report must contain, from the title and abstract through the methods, results, discussion, and supplementary material. Following PRISMA 2020 makes a review legible to peer reviewers, replicable by other researchers, and acceptable to the journals that increasingly require it.
The standard was published by Page and colleagues in the BMJ in 2021[1] as a full revision of the original PRISMA 2009 statement[2]. PRISMA 2020 added sub-items to several reporting requirements, introduced separate identification pathways for databases, registers, and other sources, and added items for protocol registration, automation tooling, and the certainty-of-evidence framework applied. This guide walks through the checklist structure, the four-phase flow diagram, the family of PRISMA extensions, and the common reporting mistakes that reviewers and methods editors flag.
Key takeaways
- PRISMA 2020 is the current standard; PRISMA 2009 is superseded for new reviews.
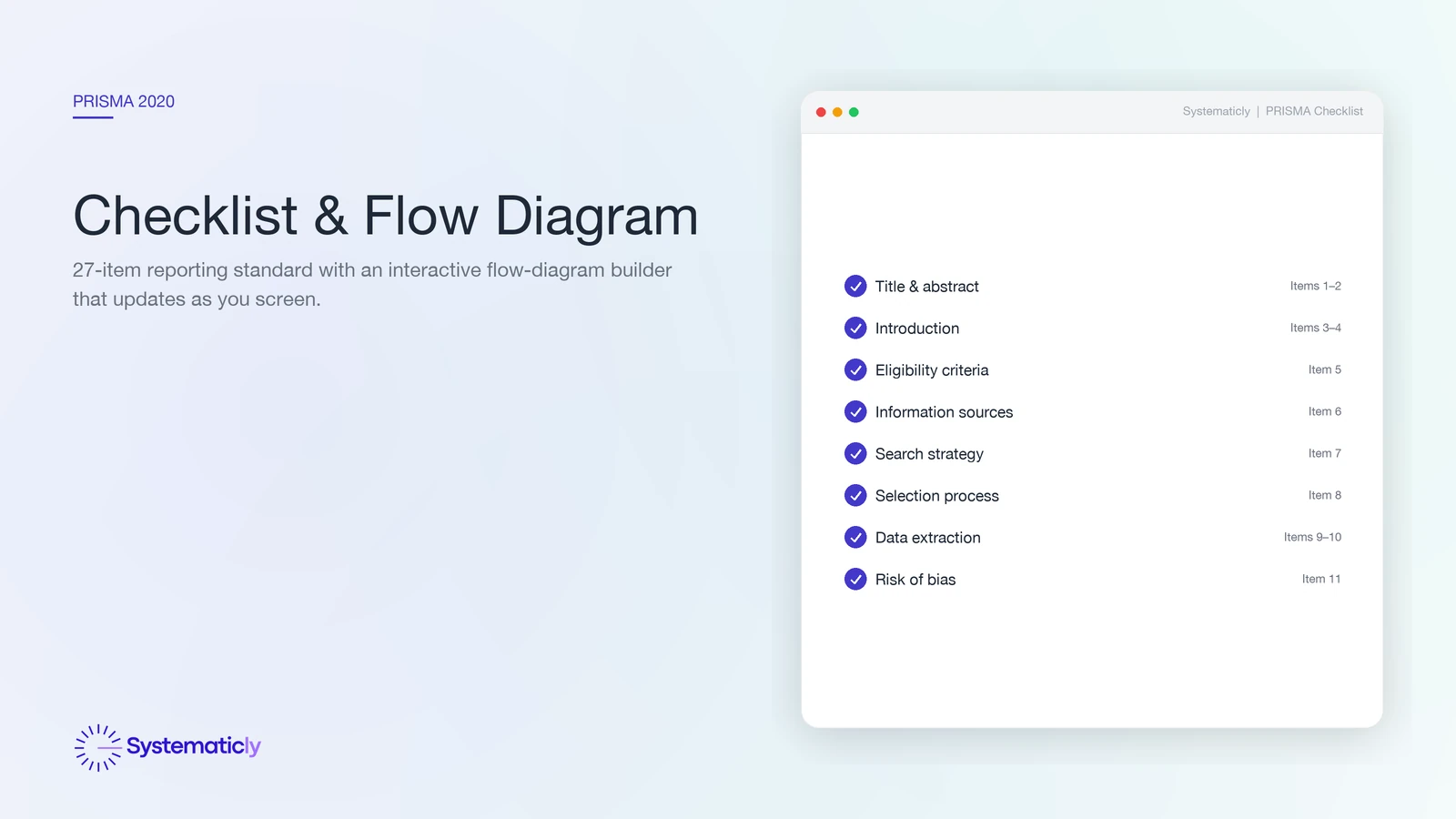
- The checklist has 27 items across title, abstract, introduction, methods, results, discussion, and other information.
- The flow diagram has four phases (identification, screening, eligibility, included) with separate columns for database and register pathways and an "other sources" pathway.
- Generate the diagram from your actual screening data; redrawing it by hand at the end is the most common source of inconsistencies.
- Use the right extension for the right review type: PRISMA-S for searches, PRISMA-NMA for network meta-analyses, PRISMA-DTA for diagnostic accuracy, PRISMA-IPD for individual-participant data, PRISMA-ScR for scoping reviews.
- Most journals now require PRISMA 2020 compliance; many request the completed checklist as a supplementary file.
What is PRISMA 2020?
PRISMA 2020 is the current minimum reporting standard for systematic reviews and meta-analyses, published by Page and colleagues in the BMJ in 2021. It specifies a 27-item checklist for the prose of a review and a four-phase flow diagram for the study-selection process. The standard is registered with the Equator Network and is referenced (or required) by most peer-reviewed health and medical journals.
The intent is straightforward: a reader should be able to take any PRISMA 2020-compliant review and reconstruct the question, the methods, the included studies, and the synthesis without having to email the authors. That standard of transparency is what allows systematic reviews to inform clinical guidelines, regulatory decisions, and health-technology assessments.
Why PRISMA reporting matters
Reporting standards exist because methodologically rigorous work that is poorly reported is, for practical purposes, the same as work that was never done. Readers cannot judge a review whose search strategy is hidden, whose exclusion reasons are not given, or whose risk-of-bias judgements are described in a single sentence. The Equator Network catalogues hundreds of reporting standards across study designs; PRISMA 2020 is the canonical one for systematic reviews.
- Journal requirement. Most major medical and health journals require PRISMA 2020 compliance, with the completed checklist often submitted as a supplementary file.
- Reproducibility. A PRISMA-compliant report contains every methodological choice another team would need to replicate the search, screening, and synthesis.
- Peer review. Methods editors and statistical reviewers use the checklist as a structured prompt for their review; missing items get flagged systematically.
- Living and updated reviews. Following PRISMA 2020 (and its update extension) makes future updates straightforward, because the original report contains everything needed to re-run the methods.
PRISMA 2020 vs PRISMA 2009: what changed
PRISMA 2009 introduced the field's first systematic-review reporting standard. PRISMA 2020 expanded it to reflect a decade of practice changes and methodological advances. The headline differences:
- Sub-items added to several checklist items. The 27-item count is the same, but several items now have explicit sub-items that ask for finer detail (search dates, search-string components, dual-screening methods, etc.).
- New identification pathway for registers and other sources. The 2020 flow diagram has separate columns for database identification, register identification, and other-source identification (citation searching, grey literature, hand-searching). The 2009 diagram lumped these together.
- Protocol-registration item. 2020 explicitly requires the protocol's registration (PROSPERO or equivalent) and any amendments to be reported.
- Automation-tooling item. 2020 asks for the automation tools used in screening, extraction, or risk-of-bias assessment to be named and described.
- Certainty-of-evidence item. 2020 requires reporting of the certainty-of-evidence framework applied (typically GRADE), with judgements for each outcome.
- Updated review extension. 2020 introduced a parallel-pathway flow diagram for updated reviews, distinguishing previous-review studies from new-search records.
In practice, the most consequential change for authors is the new identification pathway. If your search included citation tracking or grey literature, the 2020 diagram tells the reader exactly how many records came from each source and how they fared at each phase.
The 27-item checklist
The PRISMA 2020 checklist is structured by manuscript section, so it doubles as a writing prompt. The full checklist is freely available from the Equator Network and the PRISMA statement website. The structure:
- Title (1 item) — identify the report as a systematic review.
- Abstract (1 item) — structured abstract following PRISMA-Abstract.
- Introduction (2 items) — rationale and objectives (PICO/PECO/SPIDER).
- Methods (11 items) — eligibility criteria, information sources, search strategy, selection process, data-collection process, data items, study risk of bias, effect measures, synthesis methods, reporting bias assessment, certainty assessment.
- Results (8 items) — study selection (with flow diagram), study characteristics, risk of bias in studies, results of individual studies, results of syntheses, reporting biases, certainty of evidence.
- Discussion (4 items) — interpretation, limitations, implications, conclusions.
- Other information — protocol registration, support, competing interests, data availability.
The Cochrane Handbook[3] recommends completing the checklist before manuscript submission and including it as a supplementary file. Most journals now require this.
Anatomy of the flow diagram
The PRISMA 2020 flow diagram is the visual summary of the study-selection process. Four phases stack vertically: Identification (records identified from each source), Screening (records screened, records excluded), Eligibility (full-text reports assessed, full-text reports excluded with reasons), and Included (studies included in the review).
The diagram has a strict structural rule: the arithmetic at every transition must balance. If the Identification phase says 3,847 records and the Screening phase says 892 duplicates removed and 2,955 records screened, those three numbers must add up. Reviewers and editors check this; mismatches signal that the diagram was redrawn by hand rather than generated from the actual screening data. The badge in the figure above — "100% PRISMA 2020 compliant" — refers to the diagram's structural compliance, not your review's overall methodological compliance.
Identification: records, dedupe, register pathway
The Identification phase counts every record retrieved by the search, broken down by source. PRISMA 2020 distinguishes three identification pathways: database records, register records (clinical-trial registries, PROSPERO), and other sources (citation searching, hand-searching, contact with authors, grey literature). Each pathway has its own column on the flow diagram.
Deduplication is the second arithmetic step, and the diagram requires the before-dedupe and after-dedupe counts. Cross-database overlap is real and substantial: a typical clinical-question search across five databases produces a 20 to 30 per cent duplication rate. Reporting the overlap explicitly is part of PRISMA-S compliance.
Screening and eligibility: counts and reasons
The Screening and Eligibility phases each have two boxes: the records assessed and the records excluded. The Eligibility phase has a hard requirement that the full-text exclusions be reported with reasons. A line that just says "239 full-text reports excluded" without a reason breakdown will be flagged by methods reviewers and most journals.
Typical exclusion reasons at the full-text stage: wrong population, wrong intervention, wrong comparator, wrong outcome, wrong study design, duplicate publication, full text unavailable, language out of scope. PRISMA 2020 expects each reason to be reported with a count, either in the diagram or in a supplementary table referenced from the diagram.
Building the diagram as you screen
The single most common source of PRISMA-diagram errors is hand-redrawing the diagram at the end of the review from notes taken during screening. Numbers drift, exclusion reasons get rounded, and the prose stops matching the diagram. The remedy is to generate the diagram from the actual screening data, updating it as work progresses.
A live diagram during the review serves a second purpose: it gives the team a real view of progress. "1,423 of 2,955 records screened" is more useful than a screening tracker spreadsheet, because it reads against the same structural template the final report will use. When a phase completes, the diagram reflects it; when an exclusion reason is updated, the count updates with it.
PRISMA for updated reviews
Updating a previously published systematic review is more efficient than starting a new one, and PRISMA 2020 includes an update extension to cover it. The update flow diagram tracks two parallel pathways: studies from the previous review on one side and records identified by the new search on the other. The two converge into a combined screening process and a final included-studies count that the published update reports against.
Living systematic reviews use the same machinery on a continuous basis: the search is re-run on a schedule (monthly, quarterly), new records flow into the existing project, and the included-studies count updates as borderline candidates are resolved. The Cochrane Living Systematic Review framework formalises this process and is increasingly common for fast-moving evidence bases.
The PRISMA family: extensions and the Equator Network
PRISMA 2020 is the base statement, but the family of extensions covers specific review types and reporting elements. Use the right extension for the right review type. The Equator Network is the canonical catalogue of all reporting standards including PRISMA.
- PRISMA-S[4] — the search-strategy extension. Requires full search strings for every database, vendor and platform names, search dates, supplementary searching methods, and peer-review of the strategy.
- PRISMA-NMA[5] — network meta-analyses. Adds items for the network diagram, transitivity and consistency assessment, and ranking metrics (SUCRA).
- PRISMA-IPD — individual-participant-data meta-analyses. Adds items for data-sharing agreements, IPD-specific bias assessment, and harmonisation methods.
- PRISMA-DTA[6] — diagnostic test accuracy reviews. Pairs with the QUADAS-2 risk-of-bias tool.
- PRISMA-ScR[7] — scoping reviews. A 22-item checklist tailored to mapping rather than synthesising the evidence.
- PRISMA-Abstract — the structured-abstract extension. Twelve items covering background, objective, methods, results, and conclusions.
- PRISMA 2020 update extension — for updated and living reviews, with the parallel-pathway flow diagram described above.
Common reporting mistakes
Methods editors and Cochrane reviewers see the same handful of PRISMA reporting errors repeatedly. The five most common, in roughly descending frequency:
- Diagram and prose disagree. The flow diagram says 247 full-text reports assessed; the prose says 245. Always generate the diagram from the actual screening data so the numbers cannot drift.
- Full-text exclusions without reasons. "239 reports excluded" is not enough; PRISMA 2020 requires the reason breakdown. Provide it in the diagram or in a referenced supplementary table.
- Search strategy reported only for the first database. PRISMA-S requires the full search string for every database with date, vendor, and platform. Lumping these together fails the standard.
- Risk-of-bias summary missing or per-study only. RoB judgements are made per outcome (per study, per outcome). Report them at that granularity, with a domain-level summary.
- Protocol amendments not reported. Protocol amendments are allowed, but the review must list every amendment with date and rationale. Silent amendments are a transparency failure.
Frequently asked questions
Is PRISMA 2020 mandatory for systematic-review publication?
PRISMA 2020 is the de-facto standard for most peer-reviewed health and medical journals. The BMJ, JAMA, Lancet, Cochrane Library, and many others require either a completed PRISMA 2020 checklist as a supplementary file or explicit statement of compliance. Where a journal has not yet updated its instructions to authors, PRISMA 2020 still represents current best practice and reviewers will expect it.
Can I still use a PRISMA 2009 flow diagram in a 2026 paper?
Strictly, no. The PRISMA 2020 statement supersedes 2009, and the 2020 flow diagram has separate identification pathways for databases, registers, and other sources (citation searching, grey literature) that the 2009 diagram could not capture. Using the 2009 diagram in a current submission signals that the search and reporting were not aligned with the latest standard, which reviewers commonly flag.
How do I report records identified through citation tracking?
PRISMA 2020 introduced a separate identification pathway for records found via citation searching, grey literature, hand-searching of journals, and contact with authors. These are reported in a parallel column on the flow diagram ("Records identified from other sources") with their own screening, eligibility, and inclusion counts, distinct from the database identification pathway.
Does PRISMA apply to scoping reviews and other review types?
PRISMA 2020 is for systematic reviews and meta-analyses. Other review types have dedicated extensions: PRISMA-ScR for scoping reviews, PRISMA-DTA for diagnostic test accuracy reviews, PRISMA-NMA for network meta-analyses, and PRISMA-IPD for individual-participant-data meta-analyses. The Equator Network catalogues all of them.
What is PRISMA-S and when do I need it?
PRISMA-S is the 2021 PRISMA extension specifically for reporting search strategies. It requires more detail than the base PRISMA 2020 statement: full search strings for every database, vendor and platform names, search dates, supplementary searching methods, and peer-review of the strategy. PRISMA-S is recommended for any systematic review and is increasingly required by methodological reviewers.
Can I update an existing systematic review under PRISMA 2020?
Yes, and PRISMA 2020 includes a dedicated update extension. The update flow diagram tracks two pathways in parallel: studies from the previous review and records identified by the new search. Both converge into a combined screening process, and the published update reports how the new evidence has changed the conclusions.
How can a tool be "100% PRISMA 2020 compliant"?
Tooling can guarantee that the artefacts it produces (the flow diagram, the checklist) follow PRISMA 2020 structure exactly. It cannot guarantee that your review's underlying methods (search comprehensiveness, dual screening, risk-of-bias judgement) meet the standard, only that the reporting of those methods follows the format the standard requires. Read "100% compliant" claims as tooling-side, not review-side.
If you are submitting or revising a systematic review, Systematicly can take the PRISMA 2020 reporting load off the manuscript-prep phase. The platform generates the diagram from your actual screening data, exports the completed checklist, and supports the update extension for living and updated reviews. Start a free project at research.systematicly.com to try it on your protocol.
Summary
PRISMA 2020 is the current reporting standard for systematic reviews and meta-analyses, with a 27-item checklist and a four-phase flow diagram covering identification, screening, eligibility, and inclusion. The PRISMA family of extensions (PRISMA-S, PRISMA-NMA, PRISMA-DTA, PRISMA-IPD, PRISMA-ScR, the update extension) covers specific review types. Generating the diagram from real screening data, not redrawing it by hand, is the single most effective practice for avoiding reporting errors. Systematicly automates the diagram, the checklist, and the update-extension pathway so the report always matches the underlying work.
Ready to make PRISMA reporting effortless?
Systematicly auto-generates the PRISMA 2020 flow diagram from your actual screening data, exports the completed checklist, and supports living-review updates. See your first compliant diagram in minutes.
References
- Page MJ, McKenzie JE, Bossuyt PM, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71. ↑
- Moher D, Liberati A, Tetzlaff J, Altman DG. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. BMJ. 2009;339:b2535. ↑
- Higgins JPT, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, Welch VA (editors). Cochrane Handbook for Systematic Reviews of Interventions. Version 6.4. Cochrane. 2023. ↑
- Rethlefsen ML, Kirtley S, Waffenschmidt S, et al. PRISMA-S: an extension to the PRISMA Statement for Reporting Literature Searches in Systematic Reviews. Systematic Reviews. 2021;10:39. ↑
- Hutton B, Salanti G, Caldwell DM, et al. The PRISMA Extension Statement for Reporting of Systematic Reviews Incorporating Network Meta-analyses of Health Care Interventions: Checklist and Explanations. Annals of Internal Medicine. 2015;162(11):777-784. ↑
- McInnes MDF, Moher D, Thombs BD, et al. Preferred Reporting Items for a Systematic Review and Meta-analysis of Diagnostic Test Accuracy Studies: The PRISMA-DTA Statement. JAMA. 2018;319(4):388-396. ↑
- Tricco AC, Lillie E, Zarin W, et al. PRISMA Extension for Scoping Reviews (PRISMA-ScR): Checklist and Explanation. Annals of Internal Medicine. 2018;169(7):467-473. ↑